 Students tug on the tubing based on the electronegativity of the atoms they represent.3D shapes of molecules are determined by VSEPR theory.The tubing represents the bonding electrons.The rules for the molecules are as follows: I ask a couple of volunteers to model a simple molecule like HCl using a long (1-2 m) piece of tubing. After doing my traditional explanation of how polar bonds can create polar molecules using Lewis diagrams and arrows, we move to the lab portion of the classroom where we have more space. Do not proceed to schedule a custom demo unless you have already conferred with the lecture demonstrator about it.When my high school chemistry class gets to polar molecules, we run into trouble because most of the students have never heard of the word “vector” and, even though we’ve covered VSEPR theory, they don’t have the spatial reasoning abilities to see how polar bonds could cancel out or contribute to a dipole by looking at drawings on the board. The custom demos section of the website is used by UO chemistry instructors to schedule demonstrations that are not listed in the database. 
The university shall not be liable for any special, direct, indirect, incidental, or consequential damages of any kind whatsoever (including, without limitation, attorney's fees) in any way due to, resulting from, or arising in connection with the use of or inability to use the web site or the content. 
The university further disclaims all responsibility for any loss, injury, claim, liability, or damage of any kind resulting from, arising out or or any way related to (a) any errors in or omissions from this web site and the content, including but not limited to technical inaccuracies and typographical errors, or (b) your use of this web site and the information contained in this web site.the university shall not be liable for any loss, injury, claim, liability, or damage of any kind resulting from your use of the web site. The university expressly disclaims all warranties, including the warranties of merchantability, fitness for a particular purpose and non-infringement. This web site is provided on an "as is" basis. "Do not do demos unless you are an experienced chemist!" Please read the following disclaimer carefullyīy continuing to view the descriptions of the demonstrations you have agreed to the following disclaimer. Sullivan, Department of Chemistry and Biochemistry, University of Oregon, Eugene, Oregon 97403. VSEPR Worksheet Activity I and II©2018 Chemistry Education Instructional Resources, Department of Chemistry and Biochemistry, University of Oregon, Eugene, Oregon 97403. An optional activity worksheet accompanies the molecular model kits. Optional set of Power Point slides are available to accompany this active learning activity. The suggested set of compounds all contain one or more lone electron pairs around the central atom. Given a molecular formula, students draw the Lewis Structure, Identify the total regions of electron domains around the central atom, build the model of the molecule, draw a 3D representation of the molecule, identify the molecular shape, identify the bond angles.and classify the molecule as polar or non-polar. High quality plastic individual model kits for VSEPR configurations are used to enable students to build molecular models. Students visualize and represent molecules in 3-Dimensions. An optional activity worksheet accompanies the molecular model kits.Īctive Learning Approach. Given a molecular formula, students draw the Lewis Structure, Identify the total regions of electron domains around the central atom, build the model of the molecule, draw a 3D representation of the molecule, identify the molecular shape, identify the bond angles. Optional set of Power Point slides are available to accompany this active learning activity. High quality plastic individual model kits for VSEPR configurations are used to enable students to build molecular models. 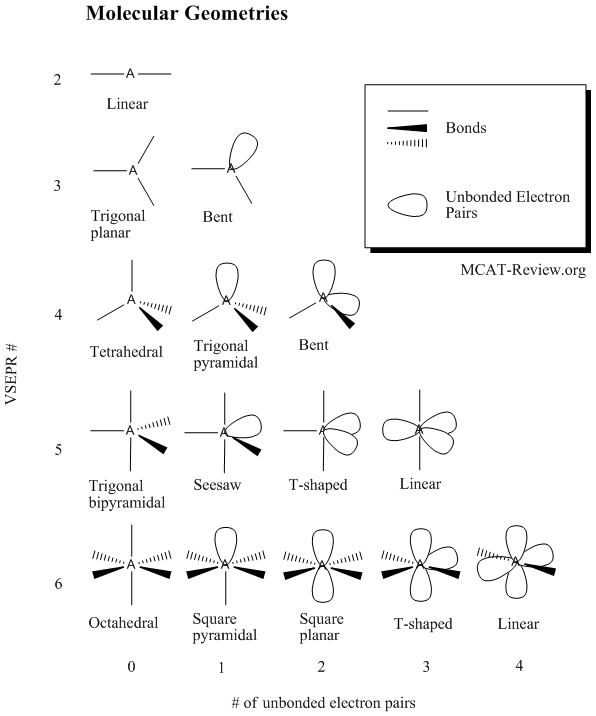
As students build the models and draw Lewis structures, more information is provided. Students do this with some (the minimum) information provided. Students assemble the six basic VSEPR molecular geometries up through the octahedral configuration, AX 6E 0. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
